
The second law of thermodynamics explains what the first law can’t, that what is in theory reversible, in practice isn’t. For example, running a video of the earth orbiting the sun looks much the same, but playing a video of an egg breaking in reverse evokes laughter. Yet by the first law, the events of egg breaking are as reversible as the earth’s orbit, so why doesn’t it happen? The second law answer is that order never increases, so eggs can’t unbreak (Figure 5.17).
Entropy is how physics describes disorder, randomness, or uncertainty. Boltzmann defined it as the number of possible microscopic states of molecules that can produce a macroscopic state, so that definition is used here. For example, when a colored gas is injected into an empty bottle, it spreads. When the gas molecules are concentrated at a point, its entropy is the number of molecule combinations that allow that, but a lot more combinations allow the spread-out state, so it has more entropy. The gas spreads because more micro-state combinations support the spread-out state, hence entropy always increases or stays constant (Figure 5.18).
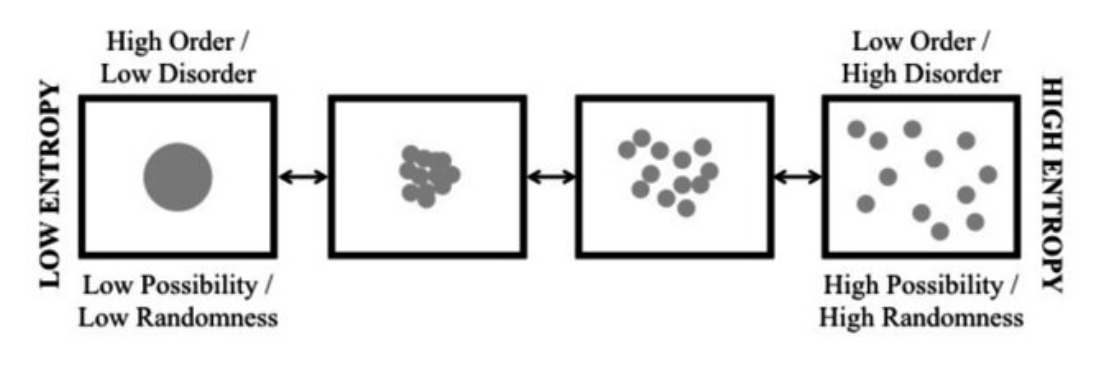
In general, entropy increases because it is more probable, so gas injected into a bottle will probably spread over time, but its molecules could by chance all move back to a point. This is unlikely but possible, so the second law is a statistical law, based on probability, not a causal law based on a force. Objects don’t have to become more disordered, but in a constantly changing world they probably will. Disorder is then probable because our world is constantly changing, just as constantly shaking a bottle makes its contents disperse.
Heraclitus compared our reality to a river that constantly changes from one moment to the next, so nothing ever stays the same. This constant flux can be attributed to the quantum law of all action (3.6.3), that quantum events explore every option, so there is always change. If the second law of thermodynamics derives from the first law of quantum theory, it is a universal law, but how then does order arise?